Public
Mecmesin Catalog
Mecmesin Certificates
Mecmesin Documents
Mecmesin Locations
Mecmesin News
Mecmesin Videos
If this is your company, CONTACT US to activate Packbase™ software to build your portal.
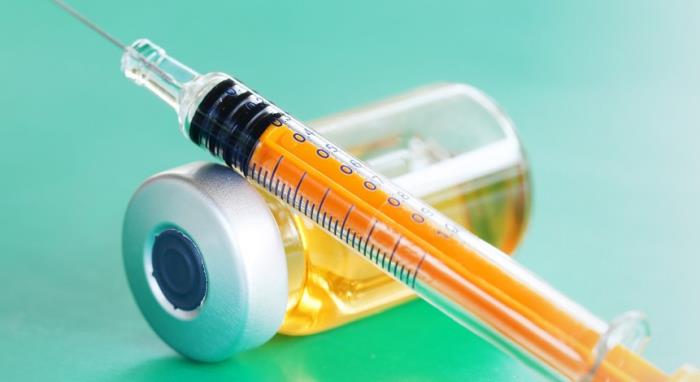

Vaccines are administered in several ways: orally; by subcutaneous (SC), intramuscular (IM) and intradermal (ID) injections and via the intranasal (IN) route. Each of these routes requires the vaccine to be carefully controlled and administered via a medical device.
All medical devices must be tested to ensure they are safe and meet stringent test standards and regulations.
Download Mecmesin's free syringes & vials whitepaper
In the whitepaper Mecmesin will provide you with an overview of the force and torque testing methods used to assess the quality and performance of syringes – from individual components through to the assembled device.
Quality through control
For SC, IM and ID routes parenteral vaccine delivery is via a sterile needle-based injector with the needle size adapted to the route, age and size of the patient.
Vaccine injections are regularly cited as a significant source of procedural pain, especially in children, leading to distress for patients and parents alike. Using force measurement equipment to test syringes by making sure that the plunger moves consistently, without stiction, reduces the chances of any jerky motion associated with inserting the needle painfully into the skin.
Quality-control checking of needle sharpness, by performing puncture-resistance testing, ensures that the needles themselves are all of a consistent standard. Performing axial load and torque tests on Luer connectors to ISO 80369 ensures needles will fit securely into the connectors and not detach.
The fear of needle stick injuries, the risk of disease transmission from needles and the need to dispose of medical waste, has led to the design of alternative delivery devices.
One needle-free approach is the intranasal multi-dose spray pump. It consists of a glass or plastic bottle which is sealed by attaching a nasal spray pump fitted with a dip tube.
The pump is actuated by pressing down the piston, creating pressure which forces liquid through the actuator to generate a spray. A valve mechanism in the metering chamber prevents backflow into the dip tube.
Proper mechanical function of spray pumps is obviously critical –that’s why force and torque tests are performed on caps, nozzles, dip tubes and spring assemblies to make sure that they meet stringent quality-control specifications.
In addition to the vaccine delivery device, packaging used to protect the vaccine also must be tested rigorously for quality control to ensure the packaging provides physical protection for the vaccines and most critically that sterile conditions are maintained up to the point of use.
It is important to understand the design of the packaging components e.g., for vials blowback features, inner neck diameters and stopper designs are important factors to consider to ensure the packaging is fit-for-purpose.
Capping equipment for vial systems play a significant role in ensuring that container closure integrity has been created from the moment the system is sealed off from the rest of the environment. Understanding the compatibility with different aluminium seals and vial systems in relation to the capping equipment can help reduce the risk of a breach in container closure integrity at the very beginning of the sealing process.
Primary vaccine packaging such as sterile containers, bottles, vials and ampoules, especially of live viral vaccines are sometimes stored at very low temperatures or are stored on dry ice during transport, to maintain stability.
These storage conditions can impact container closure integrity (CCI) e.g., rubber stoppers lose their elasticity. This loss of elasticity, coupled with inappropriate capping & crimping, can result in increased risk that the packaging seal integrity could be compromised, risking microbial contamination.
Force and torque testing systems are used to carry out a variety of tests on vaccine packaging designs, raw materials and packaging products to ensure the packaging is fit-for-purpose e.g. tensile tests on elastomeric closures; seal integrity, peel and tear properties of flexible, semi flexible and rigid vaccine packaging to test standards such as ISO-11607-1, EN 868-5 and ASTM F88 to ensure it is secure and retains its integrity and sterility but is still easy to open, plus accurate torque measurement to ensure container closure integrity (CCI).
Mecmesin has over 40 years’ experience working with medical device and medical packaging manufacturers to provide the best and most cost-effective test systems to meet their force, materials & torque testing challenges. We understand your industry-specific requirements whether it be IQ/OQ/PQ or FDA 21 CFR Part 11.
