If this is your company, CONTACT US to activate Packbase™ software to build your portal.
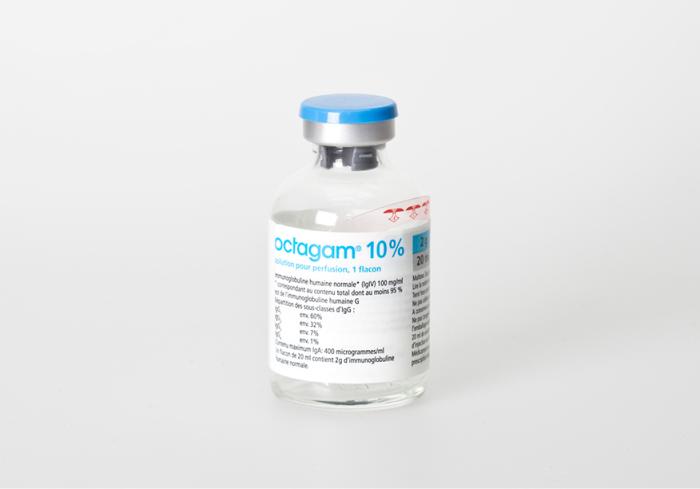

Schreiner MediPharm, a Germany-based global provider of specialty pharmaceutical labeling solutions for over 60 years, has been recognized with a first place award for its Pharma-Tac Plus hanging label. Developed jointly with Octapharma, the Pharma-Tac Plus label, with an especially robust hanger for infusion bottles, was recognized in the 'multi-process' category by the North American Tag and Label Manufacturers Institute (TLMI).
The award-winning Pharma-Tac Plus label convinced the panel of expert judges with its high-level printing technology and sophisticated label design. The label for infusion bottles combines product marking with a hanger, several pages for product information, and detachable documentation parts. The tear strength and stability of the label-integrated hanger ensure secure hanging of the infusion bottle, and its two-layered construction affords ample space for product information text in three languages, necessary for meeting regulatory requirements in multilingual countries. The label can be easily opened and reclosed, and its two detachable documentation parts for patient medical records can be post-printed with the batch number.
Many of Schreiner MediPharm's other innovative products have received awards from the North American Tag and Label Manufacturers Institute, which has been sponsoring this prestigious annual international competition to recognize the industry's best products since 1977. The panel of expert judges attaches particular importance to innovation and outstanding printing and manufacturing quality.
"The Pharma-Tac Plus label provides exceptional benefits", said Gene Dul, President of Schreiner MediPharm U.S. "The label adds value to the product and is easy to integrate into existing manufacturing processes, it lends itself to efficient and reliable use by healthcare staff, and traceability of the infusion makes an important contribution to patient safety."